Creation Could Be Left-Handed
Creation Could Be Left-HandedWhatever the right hand findeth to do, the left hand carries a watch on its wrist to show how long it takes to do it. - Ralph W Sockman by Dr David Whitehouse In 1848 Louis Pasteur discovered that some molecules can exist in two mirror-image forms, termed right or left-handed. Curiously, in living things the molecules tend to be one of these types and not a mixture of both. Amino acids, the molecular building blocks of life are all left-handed where as sugars, including deoxyribose an important component of DNA, are always right handed. By contrast, when these molecules are made in the laboratory equal numbers of the right and left-handed versions are made. Why life on the molecular level is like this is a mystery but now an international team of astronomers may have discovered why life on planet Earth is left-handed. The astronomers believe that this left-hand bias began long before our Sun was even born. At the time when our star was forming out of a vast cloud of gas and dust more than five billion years ago. Comets, mountain-sized dirty snowballs of dust and frozen gases, were formed in this Sun's birth-cloud. The left-handed molecules were incorporated into comets and found their way to Earth as they struck the young planet. Using the Anglo Australian Telescope to study a stellar nursery scientists may have discovered how the left-hand effect came about. Professor James Hough of the University of Hertfordshire, England, said: "We detected high degrees of circularly polarised light in a region of the Great Nebula in Orion where new stars are being formed. This region may well be similar to the region in which our own solar system formed." The importance of the detection of polarised light in the stellar nursery is that such light is known to destroy one of the types of mirror molecules leaving an imbalance. This is a profound discovery. Many scientists believe that life could only have developed on Earth because of the bias towards left-handed molecules. If this is so then one of the reasons we are here is because of the behaviour of light in the vast cloud of gas and dust which gave birth to our Sun billions of years ago. Source: BBC Online Thursday 30 July 1998 (Dr Whitehouse is the BBC science editor)
Describing ChiralityKelvin's Definition"I call any geometrical figure, or group of points, chiral, and say it has chirality, if its image in a plane mirror, ideally realised, cannot be brought to coincide with itself." This is the celebrated definition stated by Lord Kelvin in 1904, in his Baltimore Lectures on Molecular Dynamics and the Wave Theory of Light. This statement is universally accepted as the definition of chirality. Equivalent statements simply transpose Kelvin's definition in other contexts, such as symmetry groups. Chirality is a dichotomous propertyThe word "chiral" comes from the ancient Greek "cheir" which means "hand". The definition of chirality is something which has a mirror image, but cannot be superimposed. These two mirror images are known as "enantiomers". They are, unsurprisingly, left and right "handed". A mixture of these enantiomers in equal proportions is known as "racemic", but of course they can be separated. Examples of chiral objects in everyday life are: hands, shoes, ears. Examples of achiral objects are things like: balls, forks,hammers. Take an arbitrary "geometrical figure." Create its mirror image. Then try to match the two figures. There are only two possible outcomes: they are identical (the figure is achiral) or they are not identical (the figure is chiral). It works just like a toggle switch: the switch is either on or off. Accordingly, a chiral aminoacid like L-alanine is just as chiral as the parent compound L-phenylalanine. Kelvin's framework simply tells us that L-alanine and L-phenylalanine are not achiral. Kelvin's definition is basically the definition of achirality. Chirality is only defined negatively; the chiral objects are the objects that fail to be achiral. Chirality is a geometrical propertyKelvin himself spoke of "geometrical figures" and "groups of points" only. Thus, take a geometrical figure. Constructing its mirror image is applying the geometrical operation known as a plane symmetry. Comparing the initial object and its image proceeds by rotating or translating any one of them. Now, plane symmetry, rotations and translations are geometrical operations known as isometries. Thus, chirality is a purely geometrical property. Moreover, this geometrical property depends on a definite way of handling the objects considered: in Kelvin's framework, objects are handled through isometries. A first consequence is that chirality is not related in any particular way with chemistry or biology. Kelvin's definition applies to molecules just as it applies to knots and other mathematical constructs because they are "sufficiently geometrical" to be handled through isometries. Whereas chirality indeed plays a tremendous role in organic chemistry or biochemistry, chirality does not depend on carbon atoms properties. Can we go beyond Kelvin's definition? There is a long-standing tendency among chemists to consider some molecules "more" chiral than others. This intuition is based on a wealth of experimental evidence. Occasionally, but regularly, this idea is explicitly stated in research articles. However, it must be emphasised that Kelvin's definition definitely prohibits such statements. There lies a clear-cut and seemingly hopeless contradiction, and this contradiction originates in Kelvin's definition itself. With no way out of this contradiction, it seems that the chemist's intuition that molecules could be assigned different chiralities on a firm basis is to remain a dream. But yes, we can describe chirality. A way out of this contradiction was found in the years 95-98, and two research articles presenting this approach were published in 97 and 98. It was mathematically proved that chirality is amenable to a rich description for a large class of objects that includes molecules. This approach was called the gauge description of chirality, because it is associated with a new class of geometries called gauge geometries. It may not be clear how a geometry could allow us to go further than Kelvin's definition. Besides, what mathematicians call a geometry may not be clear to everybody. Imagine that you had a left hand transplanted in the place of your right hand. Now try and shake hands with a friend. You're in trouble! Whichever way the left hand was transplanted, your fingers would seem to be inverted. Source: mostly from chiral.com/description/research.htm
On the Other Hand: Researchers Report First Experimental Evidence of Optical Chirality in Tiny Nanoclusters of GoldA report published in the 30 March 2000 issue of the Journal of Physical Chemistry presents the first experimental evidence that tiny nanoclusters of metallic gold - assemblies containing between 20 and 40 gold atoms encapsulated by a common biomolecule - can display distinctly chiral properties. The chiral nature of the clusters, which means they exist in distinct right-handed and left-handed variations, dramatically affects the way in which they absorb polarised light. This optical effect had been predicted theoretically to occur in metal nanostructures, but Georgia Institute of Technology researchers were the first to measure it in a special class of clusters they formulated. "When clusters are prepared in this way, we see that the conduction electrons in the gold circulate in such a way as to have the unique optical effect of preferring one direction of circularly-polarised light over the other direction," explained Dr Robert L Whetten, a professor in Georgia Tech's School of Physics and School of Chemistry and Biochemistry. "The effect was enormous, which was unexpected." The gold nanoclusters are believed to be the smallest ever prepared. Dr T Gregory Schaaff, a former graduate student in Whetten's lab and now a staff scientist at the Oak Ridge National Laboratory, attached glutathione - a common sulfur-containing tripeptide - to individual gold atoms to form a gold-glutathione polymer in which the gold atoms make no direct contact with one another. The decomposition of this polymer yielded the gold clusters, which have glutathione molecules adsorbed to their surface so as to physically limit the number of metal atoms that could join together in each cluster. While measuring the properties of the clusters, Schaaf noted dramatic differences in the way the smallest clusters absorbed polarised light in the visible and near-infrared spectra. In one cluster, this circular dichroism effect exceeded 300 ppm in the yellow-green region, while in another cluster, the effect exceeded 1,000 ppm in the red and near-infrared.
Using gel electrophoresis to separate the clusters by weight, Schaaff found that certain cluster sizes dominated, with 28-atom assemblies - slightly less than one nanometer across - being the most common. The chiral properties varied by the size of the cluster, and therefore were only observed clearly when the clusters were separated by weight. Only clusters with 40 or fewer atoms displayed the intense optical properties. The optical effect changed direction as the researchers moved from one cluster size to the next, suggesting a direct correlation to the energies of the conduction electrons in the metal's outer shell. "Even though the optical absorption increases more or less monotonically here, the preferences for right-versus left-handed light changes direction from one band to another," Whetten noted. "The optical spectra are not smeared out. They each have their own distinct character, plus or minus, corresponding to the energy level." He believes the effect is related to the high level of confinement created in the conduction electrons by formation of the small clusters, though research has not yet confirmed that. A helical geometrical pattern or "tiling" of the glutathione adsorption sites (gold-sulfur bonds) could also affect the circulation of the conduction electrons. The implications and potential uses for the effect also remain to be determined. "Having this kind of a structure is a big deal in terms of the way they interact with light, and maybe the way they interact with other things that are chiral," Whetten said. "From the point of view of metallic bonding, its actually a subtle difference physically. But it makes an enormous difference in how they interact with light, and perhaps under certain circumstances, what they can do chemically." The next step in the research is to characterise the clusters to determine what causes this effect. Whetten and Schaaff want to study the internal structure of the gold clusters in addition to their interactions with the tripeptides. "We need to rigorously determine the arrangement of the atoms, to find out what aspects of the internal structure give rise to this effect," Whetten continued. "We want to understand the reasons why, out of this whole range of sizes from about 20 atoms to about 80 atoms, there are only a handful of distinct sizes that are dominating." Whetten would also like to make the new gold-glutathione clusters available to other researchers for study - and potential development of new applications. "We can make these very easily in large enough quantities to share with others interested in working with them," he said. For several years, Whetten's group has been collaborating with researchers at the University of North Carolina - Chapel Hill to study the unique electrical properties of gold nanoclusters. The new gold-glutathione clusters are of interest not only because of potential electronic applications, but because they can be used as markers in nanoprobes - their presence indicated by their unique light absorption. Glutathione is a non-protein combination of amino acids synthesized by cells to help maintain proper reduction-oxidation levels (that is, to prevent ordinary oxygen and free radicals from "burning up" the fragile biomolecules in the living cell). Its importance to this work stems from its unique molecular structure that tends to favour the formation of small gold nanoclusters. The research was sponsored by the US National Science Foundation and the Georgia Tech Foundation. Source: gtresearchnews.gatech.edu from the Research News & Publications Office of the Georgia Institute of Technology 430 Tenth Street, NW, Suite N-116 Atlanta, Georgia 30318 USA
Knot a Problem!(If no knot appears below, try reloading or refreshing the page as it only reties itself 10 times before disappearing.)
We all know what a knot is and what a knot is not. At least we think we do. A knot is what our shoelaces end up in when we try to pull the end of that elegant bow we made out of the lace. For mathematicians, the definition of a knot has to be more precise. Rather than bothering to define a knot, mathematicians ask the more useful question: when are two knots equivalent topologically? Suppose we are given two simple closed curves in three dimensions. The branch of mathematics called knot theory provides us will tools to check whether one knot can be continuously transformed into another. A very special case of this problem is to determine whether a knot is equivalent to its mirror image. If we hold some knots up in front of a mirror we can see regard the mirror image as another knot. When it is possible to continuously transform a knot into its mirror image, the knot is said to be achiral. Otherwise, the knot is said to be chiral. The animation above shows that the figure eight knot seen at the beginning can be transformed into its mirror image, which appears at the end. A wonderful and accessible book on Knot Theory is Colin C Adams' The Knot Book, published by Freeman and Company. It requires no advanced mathematics but looks at some deep problems involving knots. Source: stats.uwaterloo.ca University of Waterloo, Department of Statistics and Actuarial Science, Waterloo, Ontario, Canada
Scientists Can Be Less than TactfulPosted by dr_syed_ameen2000 on 8 February 2001:
Posted by Frink on 28 March 28 2001:
Posted by DickT on 9 February 2001:
Source: superstringtheory.com
For articles on bacteria, centrioles, chairs, nebulae, asteroids, robots, memory, chirality, pain, fractals, DNA, geology, strange facts, extra dimensions, spare parts, discoveries, ageing and more
click the "Up" button below to take you to the Index for this Science section. |
 Animals
Animals Animation
Animation Art of Playing Cards
Art of Playing Cards Drugs
Drugs Education
Education Environment
Environment Flying
Flying History
History Humour
Humour Immigration
Immigration Info/Tech
Info/Tech Intellectual/Entertaining
Intellectual/Entertaining Lifestyles
Lifestyles Men
Men Money/Politics/Law
Money/Politics/Law New Jersey
New Jersey Odds and Oddities
Odds and Oddities Older & Under
Older & Under Photography
Photography Prisons
Prisons Relationships
Relationships Science
Science Social/Cultural
Social/Cultural Terrorism
Terrorism Wellington
Wellington Working
Working Zero Return Investment
Zero Return Investment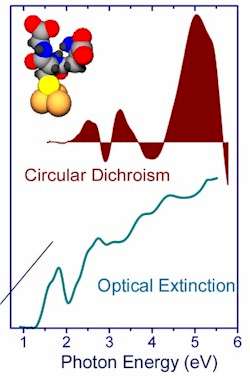 These optical measurements suggest that the clusters have a helical structure that Whetten compared to the stripes on
a candy cane or a barbershop pole. "We had to double-check our instruments and repeat the measurements a number of times because the effect was enormous," he said. "This effect is comparable
to what is seen in naturally-helical structures. Such effects had not previously been measured in metal-cluster compounds and it's kind of a shock that small metals might prefer to have a helical
structure."
These optical measurements suggest that the clusters have a helical structure that Whetten compared to the stripes on
a candy cane or a barbershop pole. "We had to double-check our instruments and repeat the measurements a number of times because the effect was enormous," he said. "This effect is comparable
to what is seen in naturally-helical structures. Such effects had not previously been measured in metal-cluster compounds and it's kind of a shock that small metals might prefer to have a helical
structure."