Here's a Clue: Living, Dead
Cell's Recycling May Yield Clues to AgeingThe most exciting phrase to hear in science, the one that heralds new discoveries, is not "Eureka!" but "That's funny..." - Isaac Asimov
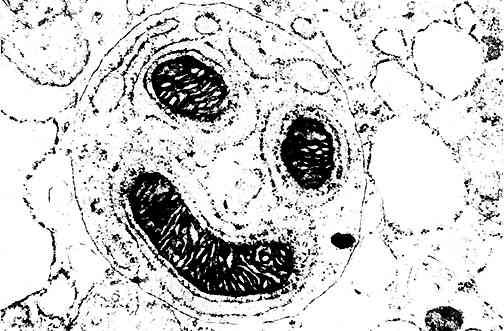
Mitochondrion (this one looks fairly happy...) New York - Scientists have identified a whirling "dervish-like" structure within cells that helps to recycle existing compounds into ATP, the molecule that stores the energy needed for life. They believe the discovery could point to new methods of slowing the ageing process. The structure is "one of the most complex molecules ever revealed," said Dr Peter Pedersen, a researcher at Johns Hopkins University in Baltimore, Maryland, and co-author of a study published in the September 1998 issue of the Proceedings of the National Academy of Sciences. Scientists have long understood that cellular energy production occurs in the mitochondria, capsule-shaped "power plants" found in the cell. Still, the exact mechanisms behind this process have remained unclear. The Johns Hopkins team believes that they may have cleared up some of that mystery. They focused their efforts on tiny, mushroom-shaped molecules found within the mitochondria. These molecules are enzymes called adenosine triphosphate synthase, which make ATP. The ATP synthase enzyme is a relatively complex structure "almost 6 times larger than the blood molecule hæmoglobin," Pedersen explained. Examination of ATP in rat liver cells revealed that the "stem" portion of the mushroom-shaped molecule revolves, dervish-like, under the "cap."; As the stem rotates, it creates enough energy to turn on the ATP-synthesis "factory" located within the cap. The ingredients necessary for the formation of ATP are constantly recycled within this cap, according to the study authors. When ATP synthesis is complete, the cap disengages from the stem, allowing ATP to be used as an energy source within the cell. The recycling of ATP helps explain the body's ability to function without an enormous daily intake of food. Without this recycling process, "people would have to produce more than half their body weight in ATP every day to meet their energy needs," Pedersen explained. He noted that "free radicals" - the negative by-products of metabolism - could potentially damage ATP synthesis at the molecular level. Scientists have long believed that the gradual accumulation of free radical damage within cells plays a key in both disease formation and the ageing process. "If ATP synthase is a site of free radical damage," Pedersen speculated, "that could explain why we become less energetic with age." Antioxidant compounds (including vitamins C and E) are thought to "mop up" free radicals and improve cellular health. Pedersen said that a better understanding of the cell's energy production systems could someday help researchers develop agents that could repair damage caused by free radicals. "Now that we know the molecular structures (involved in energy production), we can pinpoint damaged regions if they occur," he explained. From the Proceedings of the National Academy of Sciences 1998 95:11605-11070 Source: Reuters Friday 25 September 1998 See also:
Living Dead: Ants and Infertile Humans Are Not Alive, but Parasitic DNA Is, According to a New, Universal Definition of LifeWomen over 50 already form one of the largest groups in the population structure of the western world. As long as they like themselves, they will not be an oppressed minority. In order to like themselves they must reject trivialization by others of who and what they are. A grown woman should not have to masquerade as a girl in order to remain in the land of the living. - William Golding Ants and infertile humans are not alive, but parasitic DNA is. That's the view of a Polish researcher who has proposed a new, universal definition of life. He claims it will lay to rest arguments about what is and isn't alive, and might offer insights into when life on Earth got started. And if we ever find something that looks like life on another planet, his definition could help us settle whether it's alive or not. Bernard Korzeniewski of the Institute of Molecular Biology at Jagiellonian University in Kraków, Poland, says that definitions of life usually list the attributes an organism must have, such as genes, a certain level of complexity, the ability to reproduce and evolve, and so on. But this, he says, merely describes life rather than providing a useful way of deciding what's alive and what's not. So Korzeniewski set out to formulate a fundamental definition of life, "which would apply not only to life presently existing on our planet but also to the first living organisms on Earth, as well as to life-like phenomena existing presumably on other planets in the Universe", as he puts it. His definition is this: "A network of inferior negative feedbacks subordinated to a superior positive feedback." In other words, it's a system that tries to regulate itself to preserve its identity. Defining FunctionIt might not sound like much, but Korzeniewski is convinced that this broad formulation - which he calls a cybernetic definition of life - pinpoints the defining function of life, rather than listing its attributes. "In spite of the enormous diversity of life forms, the most fundamental principles of biochemical construction and function are astonishingly alike," he says. Under his definition, sterile worker ants are not alive because they rely on others to reproduce and preserve their own identity. And although it would be anathema to most people, he says the same applies to infertile humans. But an ant colony is a living system, according to Korzeniewski, because individuals work together to preserve the colony as a whole. "An ant is alive in the manner, say, a liver or a heart is - only as a part of some bigger system," he says. Individual ants may be complex systems, but complexity is not unique to life - just look at computers. Parasitic DNASimilarly, viruses, cancers and parasitic DNA that has no function but gets passed down the generations, all count as alive. All are capable of producing regulatory enzymes that ultimately help them reproduce. But prions and viroids - disease-causing particles of RNA smaller than a virus - do not have this ability, and should not be considered alive. One reason why life is such a great mystery is that no one has yet been able to define it, says Mark Bedau, a philosopher of biology at Reed College in Portland, Oregon. Bedau welcomes the generality of the new definition. "But the test of a good definition of life is whether it can settle ambiguous cases," he says. Korzeniewski says his definition also pinpoints when life emerged. He believes life began with RNA - the first molecules capable of perpetuating their identity through replication. More at: Journal of Theoretical Biology (vol 209, p 275) Source: newscientist.com 16 May 2001 Correspondence about this story should be directed to letters@newscientist.com
-------- Original Message --------
Source: physforum.com Suggesting,
The Origin of Life"On this single planet called Earth, there co-exist (among countless other life forms), algae, beetles, sponges, jellyfish, snakes, condors, and giant sequoias. Imagine these 7 living organisms lined up next to each other in one-place. If you didn't know better, you'd be hard-pressed to believe that they all came from the same universe, much less the same planet." - Neil deGrasse Tyson The search for life in the universe begins with a deep question: what is life? Astrobiologists will tell you honestly that this question has no simple or generally accepted answer. Not much use to say that we'll know it when we see it. No matter what characteristic we specify to separate living from nonliving matter on Earth, we can always find an example that blurs or erases this distinction. Some or all living creatures grow, move, or decay, but so too do objects that we would never call alive. Does life reproduce itself? So does fire. Does life evolve to produce new forms? So do certain crystals that grow in watery solutions. We can certainly say that you can tell some forms of life when you see them - who could fail to see life in a salmon or an eagle? - but anyone familiar with life in its diverse forms on Earth will admit many creatures will remain entirely undetected until the luck of time and the skill of an expert reveal their living nature. Since life is short, we must press onward with a rough-and-ready, generally appropriate criterion for life. Here it is: Life consists of sets of objects that can both reproduce and evolve. We shall not call a group of objects alive simply because they make more of themselves. To qualify as life, they must also evolve into new forms as time passes. This definition therefore eliminates the possibility that any single object can be judged to be alive. Instead, we must examine a range of objects in space and follow them through time. This definition of life may yet prove too restrictive, but for now we shall employ it. As biologists have examined different types of life on our planet, they have discovered a general property of earthlife. The matter within every living earth creature mainly consists of just 4 chemical elements: hydrogen, oxygen, carbon, and nitrogen. All the other elements together contribute less than 1% of the mass of any living organism. The elements beyond the big 4 include small amounts of phosphorus, which ranks as the most important, and is essential to most forms of life, together with still smaller amounts of sulfur, sodium, magnesium, chlorine, potassium, calcium, and iron. But can we conclude that this elemental property of life on earth must likewise describe other forms of life in the cosmos? Here we can apply the Copernican principle in full vigour. The 4 elements that form the bulk of life on earth all appear on the short list of the universe's 6 most abundant elements. Since the other two elements on the list, helium and neon, almost never combine with anything else, life on earth consists of the most abundant and chemically active ingredients in the cosmos. Of all the predictions that we can make about life on other worlds, the surest seems to be that their life will be made of elements nearly the same as those used by life on earth. If life on our planet consisted primarily of four extremely rare elements in the cosmos, such as niobium, bismuth, gallium, and plutonium, we would have an excellent reason to suspect we represent something special in the universe. Instead, the chemical composition of life on our planet inclines us toward an optimistic view of life's possibilities beyond earth. The composition of life on earth fits the Copernican principle even more than one might initially suspect. If we lived on a planet made primarily of hydrogen, oxygen, carbon, and nitrogen, then the fact that life consists primarily of these four elements would hardly surprise us. But earth is mainly made of oxygen, iron, silicon, aluminium, and iron. Only one of these elements, oxygen, appears on the list of life's most abundant elements. When we look into earth's oceans, which are almost entirely hydrogen and oxygen, it is surprising that life lists carbon and nitrogen among its most abundant elements, rather than chlorine, sodium, sulfur, calcium, or potassium, which are the most common elements dissolved in seawater. The distribution of the elements in life on earth resembles the composition of the stars far more than that of earth itself. As a result, life's elements are more cosmically abundant than earth's - a good start for those who hope to find life in a host of situations. Neil De Grasse Tyson is Frederick P Rose Director, Hayden Planetarium, American Museum of Natural History (since 1996); Visiting Research Scientist, Department of Astrophysics, Princeton University (since 1994). He writes a monthly column called "Universe" for Natural History magazine and is the author of several books, including One Universe: At Home in the Cosmos (2000) and The Sky is Not the Limit: Adventures in an Urban Environment (2000). His most recent work is the book (published by WW Norton & Co) and NOVA PBS four-part series, Origins. This is excerpted from chapter 15, titled The Origin of Life on Earth. Source: space.com 30 November 2004 Also see:
For articles on bacteria, centrioles, chairs, nebulae, asteroids, robots, memory, chirality, pain, fractals, DNA, geology, strange facts, extra dimensions, spare parts,
discoveries, ageing and more click the "Up" button below to take you to the Table of Contents for this Science section. |
 Animals
Animals Animation
Animation Art of Playing Cards
Art of Playing Cards Drugs
Drugs Education
Education Environment
Environment Flying
Flying History
History Humour
Humour Immigration
Immigration Info/Tech
Info/Tech Intellectual/Entertaining
Intellectual/Entertaining Lifestyles
Lifestyles Men
Men Money/Politics/Law
Money/Politics/Law New Jersey
New Jersey Odds and Oddities
Odds and Oddities Older & Under
Older & Under Photography
Photography Prisons
Prisons Relationships
Relationships Science
Science Social/Cultural
Social/Cultural Terrorism
Terrorism Wellington
Wellington Working
Working Zero Return Investment
Zero Return Investment